|
Answer-> as the electron falls, it releases energy as lightħ4 State how bright-line spectra viewed through a spectroscope can be used to identify the metal ions in the salts used in the flame tests. Answer-> an electron is the higher, 4th energy level when it could occupy a lower energy levelħ3 Explain, in terms of electrons, how a strontium salt emits colored light. The evaluation of the average size of gold nanoparticles can be achieved by fitting their UV-vis spectra using the Mie model for. Each metallic salt emits a characteristic colored light in the flame.ħ2 Explain why the electron configuration of 2-7-1-1 represents a sodium atom in an excited state. During a flame test, a metallic salt is heated in the flame of a gas burner. A technician investigates what colors are produced by the metallic salts by performing flame tests. The outer darts of the flame are tinged with emerald-green. 
The color can be used to detect halides by using copper oxide moistened with test solution. High abundances of HCN and C2H2 are caused in the model by stellar X-ray irradiation of the gas around the inner rim.Base your answers to questions 72 through 74 on the information below and on your knowledge of chemistry.įireworks that contain metallic salts such as sodium, strontium, and barium can generate bright colors. The copper flame color is dependent on the presence of halide (I, F, Br, or Cl). The spatial structure of the line emitting regions of the different molecules is discussed. We develop a disc model with a slowly increasing surface density structure around the inner rim that can simultaneously fit the spectral energy distribution, the overall shape of the JWST spectrum of EX Lupi, and the main observed molecular characteristics in terms of column densities, emitting areas and molecular emission temperatures, which all result from one consistent disc model. Nowadays, the accurate atomic data (energy levels, transition rates and oscillator strengths) of strontium have high astrophysical interest since the announcement of its presence in the merger of two neutron stars 1, 2 and in the Y-band spectroscopy of Galactic dwarf stars where the strontium-86, -87, and -88 isotopes have significant presence in these spectra 3,4,5,6. Table 1: Name test Flame Color Metallon (Cations) Calcium (Ca) Copper (Cu) Lithium (LI) Potassium (K) Sodium (Na) Strontium (Sr') Unknown (firework) Red Table 2: Bright Line Spectra Metallon (Cations) Line Spectra Calcium (Ca) wavelength in nanometers Copper (Cu) 311 wavelengths anometers Lithium (LI. The Sr I atomic line in the measured spectrum was found to have an apparent line width of 0.82-nm at full-width-half-maximum. We calculate infrared line emission spectra between 5 and 20 mic by CO, H2O, OH, CO2, HCN, C2H2 and H2, including lines of atoms and ions, using our full 2D predictions of molecular abundances, dust opacities, gas and dust temperatures. We apply our revised 2D models to the protoplanetary disc around the T Tauri star EX Lupi in quiescent state. We show that these assumptions are crucial for the determination of the gas heating/cooling rates and discuss how they affect the predicted molecular concentrations and line emissions. 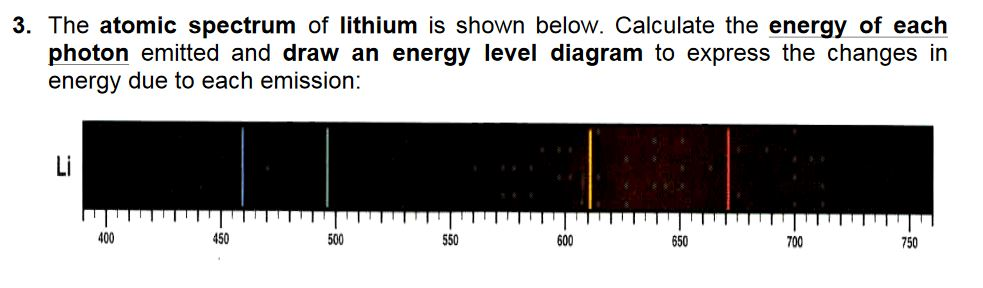
We develop a new line escape probability method for disc geometries, a new scheme for dust settling, and discuss how to apply UV molecular shielding factors to photorates in 2D disc geometry. Woitke and 5 other authors Download PDF Abstract:We introduce a number of new theoretical approaches and improvements to the thermo-chemical disc modelling code ProDiMo to better predict and analyse the JWST line spectra of protoplanetary discs. Download a PDF of the paper titled 2D disc modelling of the JWST line spectrum of EX Lupi, by P. We apply our revised 2D models to the protoplanetary disc around the T Tauri star EX Lupi in quiescent state.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
